Chemists in the US have taken an unconventional approach to carbon cross-coupling and in doing so have potentially opened the door to the rapid and efficient synthesis of a wide range of organic compounds.
Cross-coupling reactions are dominated by joining nucleophilic organoboronic acids with electrophilic organic halides, mediated by a transition metal catalyst. However, the library of commercially available organoboron compounds - and other nucleophilic carbon substrates used in this type of synthesis - is limited, and the compounds themselves can be unstable. The means that often they need to be synthesised as and when they are needed - making the process more time consuming and expensive.
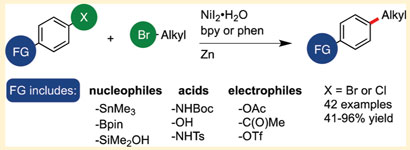
Now, Daniel Weix's team at the University of Rochester in New York has taken a different tack. Rather than coupling a nucleophile with an electrophile, the researchers link two electrophiles. 'They are typically more stable and are more commercially available than nucleophiles, so we thought it would be attractive if we could conceive of practical ways to couple two electrophiles,' says Weix.
 |
| The new technique should make a wider range of commercial reagents available for cross-coupling |
The team showed that it is possible to selectively couple aryl bromides and chlorides with alkyl bromides in high yields in a reaction catalysed by nickel in the presence of zinc.
Significantly, the reaction tolerates a wide range of functional groups on the aryl bromide. The precise mechanism of the reaction remains unclear and is being elucidated, Weix says. 'We have good evidence that we are not making an organozinc reagent in the flask. We think that the key concept to these reductive couplings is that we are putting electrons into the catalyst and letting the catalyst mediate the reaction. Zinc is the source of electrons.'
Weix believes that these electrophile-electrophile couplings could prove useful for discovery chemists. 'Chemists are often tasked with making derivatives of a core structure. If you first have to make organometallics, this can become quite a chore. The fact that there are so many more organic halides that are commercially available and easier to access could make this approach attractive.'
Vy Dong, who runs a team specialising in organic synthesis and transition metal catalysis at the University of Toronto in Canada, believes that the work has promise. 'The Weix reductive coupling is an exciting breakthrough and practical advance in cross-coupling chemistry,' she says. 'The approach provides a new way for us to think about making carbon-carbon bonds, a way that complements both traditional cross-coupling using organometallic reagents and emerging oxidative cross-coupling using C-H bonds.'
Source: Royal Society of Chemistry
No comments:
Post a Comment